To :
Faculty of Electronic and Informatics Engineering
Harvard University
Dear Dean on Electronic and Informatics Engineering of Harvard University, I have a recommendation about my student at Senior High School 3 Bandung. His name is Aira Thalca. I have known him since his arrival at Senior High School 3 Bandung. He is 17 years old, he is the first son of two siblings. His hobby is learn something new or create some project that includes programming. He has a great ability at mathematics and programming. A lot of achievement has been generated, in either city, province, or national levels. Beside that, he has a great ability in sports too, such as football and volleyball.
He is a diligent guy. He has a lot of achievement from the 1st grade. He has around 20-30 trophies in his home. Almost all of them is from Math and Science. But, some trophies won from Volleyball too. The most interesting achievement he got is when he was on JHS 1 Cimahi, He competes with Senior High School student in Math competition and got a 2nd place.
His dream is to be a programmer on Google or company in the field of gaming and someday make his own company and became success as Steve Jobs or Bill Gates.
I know him very well as my student, he is a diligent person and also smart, and also he is very interested in learning about programming. He can make some programming. He learned it just before January 2019. even so, he had attended a computer Olympics up to the provincial level.
Overall, I've no doubt that he will definitely succeed in her future academic because of his talent, and his high interest and curiosity within the realm of learning. I've no doubt that he will make a great impact on the programming world and I'm certain that he'll make an outstanding contribution to your scholarship program. I highly recommend of Aira. If you have further question, please don't hesitate to contact me.
Muhamad Fikri Nurohman
Mathematics Teacher
Senior High School 3 Bandung
Muhamad Fikri Nurohman
Selasa, 14 Mei 2019
Senin, 29 April 2019
THIS IS BALI ~~ MY TUR BUDAYA STORY
1st day
The first day of Tur Budaya 2019 I came to Husein airport at around 06:00 WIB. Before that, I had breakfast first with my family on my house. After breakfast, I said goodbye. This is my first time flight. A lot of negative feeling on my mind, but all of my family said everything would be alright...
Just around 9.30 a.m., I did a boarding pass, put the suitcase into the luggage and then around 10.15 I flight. Skip the flight story cos nothing to tell HEHE..
We arrived at the airport Ngurah Rai Bali around 12.00 WITA. After that I took the suitcase and headed for bus number 2, my class bus. First one should visit favorite destination in Bali is Tanah Lot, which is I often hear some story about it, and now it's my turn to tell you a story.
Well, Tanah Lot is a big coast, a lot of big wave in the afternoon. OMG I forgot something to tell.
So, my tour guide name is Bo Komang. On my first arrival in the bus, she teaches us some Bali Language, and the difference according to my city, Bandung. She also told that Tanah Lot is a holy place. Balinese believe in the existence of a holy snake. The holy snake is always in a silence position. Some tourist get to the holy snake place and watched it. But I don't too interested of it, I don't know why, maybe because of the beauty of Tanah Lot amaze my brain. We take a lot of photo there.
Finally tour continue to a restaurant in Jimbaran. At first we were take a photo of there then eat. But suddenly the rain fell. We went straight to the bus and went to the hotel to have a rest. The hotel is a guest house.
Just around 9.30 a.m., I did a boarding pass, put the suitcase into the luggage and then around 10.15 I flight. Skip the flight story cos nothing to tell HEHE..
We arrived at the airport Ngurah Rai Bali around 12.00 WITA. After that I took the suitcase and headed for bus number 2, my class bus. First one should visit favorite destination in Bali is Tanah Lot, which is I often hear some story about it, and now it's my turn to tell you a story.
Well, Tanah Lot is a big coast, a lot of big wave in the afternoon. OMG I forgot something to tell.
So, my tour guide name is Bo Komang. On my first arrival in the bus, she teaches us some Bali Language, and the difference according to my city, Bandung. She also told that Tanah Lot is a holy place. Balinese believe in the existence of a holy snake. The holy snake is always in a silence position. Some tourist get to the holy snake place and watched it. But I don't too interested of it, I don't know why, maybe because of the beauty of Tanah Lot amaze my brain. We take a lot of photo there.
Finally tour continue to a restaurant in Jimbaran. At first we were take a photo of there then eat. But suddenly the rain fell. We went straight to the bus and went to the hotel to have a rest. The hotel is a guest house.
2nd day
My second day in Bali starts. I wake up in the quite morning. Quickly I got my breakfast and took a bath at the hotel. The first destination is Tanjung Benoa. There, I visit to Turtle Beach. Besides there are many turtles, there are also other animals there. then I also played water sport on Tanjung Benoa, flying on the sea with flying fish, in the middle of the sea waves with donuts and it was fun. My friends, on a community called Bali 17 also played a football beach game. Indeed, the temperature was so hot, but it's alright. Everything's good and we enjoy the football. We had lunch at Tanjung Benoa too.
Okay, the second destination on this 2nd day is Pandawa Beach. According to our tour guide, formerly the Pandawa beach was a hidden beach, but after the cliff was made, road access was made. the beach becomes very famous because the view are very beauty. On the cliffs of Pandawa beach carved by several Hindu statues, We took a photos of them there. Pandawa is as hot as Tanjung Benoa. I think every beach is just like that, especially in Bali. A lot of tourists sunning themselves. On their mind, the purpose of sunburn in the beach is to prove to their friends that they have been in a beach.
When entering the prayer time we pray in all the places of worship religion namely Pujamandala. There are 5 place of worships. A mosque, temples, monasteries, and 2 churches. So we pray at that place. It is a beautiful place. We saw very high tolerance in Bali.
After that I visited Garuda Wisnu Kencana ,the largest statue in the world defeat the statue of Liberty. This statue was inaugurated on September 2018. There are a lot of statues, not only the biggest one, but also a middle and small in size. I also ride a bike for IDR 20k and had a lot of fun with my friends.
After that, in the evening, I watched the Devdan show, which is a show of all Indonesian culture from Sabang to Merauke which is very fascinating. Then rest at the hotel. A funny moments happened. So, my friends and I order some pizza. But the order destination was false, we order from Bandung to Bandung HAHAHA. But we have paid for the pizza, so all clear. We order for the second time, and it's still got some mistake. We order the pizza to Quest Hotel Kuta, even though we are in Quest Hotel Denpasar. Luckily, Kuta is not too far from Denpasar, and we still get the pizza we have ordered.
Okay, the second destination on this 2nd day is Pandawa Beach. According to our tour guide, formerly the Pandawa beach was a hidden beach, but after the cliff was made, road access was made. the beach becomes very famous because the view are very beauty. On the cliffs of Pandawa beach carved by several Hindu statues, We took a photos of them there. Pandawa is as hot as Tanjung Benoa. I think every beach is just like that, especially in Bali. A lot of tourists sunning themselves. On their mind, the purpose of sunburn in the beach is to prove to their friends that they have been in a beach.
After that I visited Garuda Wisnu Kencana ,the largest statue in the world defeat the statue of Liberty. This statue was inaugurated on September 2018. There are a lot of statues, not only the biggest one, but also a middle and small in size. I also ride a bike for IDR 20k and had a lot of fun with my friends.
After that, in the evening, I watched the Devdan show, which is a show of all Indonesian culture from Sabang to Merauke which is very fascinating. Then rest at the hotel. A funny moments happened. So, my friends and I order some pizza. But the order destination was false, we order from Bandung to Bandung HAHAHA. But we have paid for the pizza, so all clear. We order for the second time, and it's still got some mistake. We order the pizza to Quest Hotel Kuta, even though we are in Quest Hotel Denpasar. Luckily, Kuta is not too far from Denpasar, and we still get the pizza we have ordered.
3rd day
The third day in Bali as usual begins with breakfast. Today's first agenda is to visit Tirta Empul, a source of water that is considered sacred. There are several Traditional ceremonies are also held here. Besides that a lot tourists who take a bath or just wash their faces at this water source, because the rumors can make it easier Mate is ageless, and treats diseases.
So the night we are waiting comes. Everyone dress hippie, sing, play together and there are many doorprizes given, namely shopping vouchers, up to smartphones. At the end, we watched video which is displayed by Movie Production 3. The event ends in 00.00 WITA. We are so tired, return to the hotel and go to bed.
The journey continues towards Kintamani for lunch there. Actually we are presented with beautiful mountain views, however Unfortunately the weather is cloudy so the scenery is invisible. My favorite dish in Kintamani is the Satai. It's very delicioous. I add my satai for triple.
After that the journey continues to the village Penglipuran. That is a village that is famous as one of the best villages in the world, along with 2 other villages in India and Netherlands, but unfortunately when it was just until the rain fell. Well, I don't care with the weather. It's Bali bro. I persuade my friends to continue the journey in walking along the village, although it's rainy. A big rain. After that we return to the bus in a wet condition. We went back to the hotel to prepare our makrab "La boheme" in the hotel ballroom in Nirmala.So the night we are waiting comes. Everyone dress hippie, sing, play together and there are many doorprizes given, namely shopping vouchers, up to smartphones. At the end, we watched video which is displayed by Movie Production 3. The event ends in 00.00 WITA. We are so tired, return to the hotel and go to bed.
4th day
The fourth day as usual begins with breakfast at the hotel. But there is something different today, we are directly packing and checking out of the hotel. OMG it's the end of the journey. I will absolutely missed the hotel, for every moment we have there, for the crazy moments, for the deep sleep, and many more.
After clearing all the items and entering them into the bus, the journey continues to Kuta beach. We headed to Kuta Beach using a shuttle, there we went directly to the beach and were surprised by the carcass mice after that we immediately moved away from the beach and wandered around there. Not much thing we can do in Kuta Bali. I just watched some wave flows though my direction. As time went, the whether went hot and hot. So, my friend and I moved to a minimarket, and as usual in Bali. The minimarket sell an alcoholic drink. But we don't shocked. And weturned to the bus and shop for souvenirs in Krishna, and eating chicken geprek which is super spicy.
We continue to Bedugul. Bedugul is a beautiful temple I saw for the last time in Bali, because it's the final destination. Unlike usual, the temperature was so cold and the air are very fresh. It's because Bedugul located in a highland surrounded by mountains. Enjoy the air.
When night comes, we continued the journey back to Bandung, crossing using a ship from Gilimanuk port to Ketapang port on midnight.
After clearing all the items and entering them into the bus, the journey continues to Kuta beach. We headed to Kuta Beach using a shuttle, there we went directly to the beach and were surprised by the carcass mice after that we immediately moved away from the beach and wandered around there. Not much thing we can do in Kuta Bali. I just watched some wave flows though my direction. As time went, the whether went hot and hot. So, my friend and I moved to a minimarket, and as usual in Bali. The minimarket sell an alcoholic drink. But we don't shocked. And weturned to the bus and shop for souvenirs in Krishna, and eating chicken geprek which is super spicy.
We continue to Bedugul. Bedugul is a beautiful temple I saw for the last time in Bali, because it's the final destination. Unlike usual, the temperature was so cold and the air are very fresh. It's because Bedugul located in a highland surrounded by mountains. Enjoy the air.
When night comes, we continued the journey back to Bandung, crossing using a ship from Gilimanuk port to Ketapang port on midnight.
5th day
The fifth day begins with the dawn prayer and take a bath in Bromo restaurant after that Breakfast at the restaurant then travel continuing, crossing to Madura Island past the suramadu bridge. Under the suramadu bridge there is a unique phenomenon that is a border in the water the sea which is fresh water and salt water cannot be united. then on Madura Island we bought ball Madura there is a very tasty cracker ball. After that we returned to Surabaya to have lunch at Wulan warung restaurant. After that we headed Juanda station for trains 12 hours to bandung. On the train we play cards, tell stories and have dinner there. After that we rest .The sixth day is the last day of the trip from Tur Budaya to Bali. Arriving at the station Bandung, We carry all our items including souvenirs and return to their homes each safely then rest.
Senin, 15 April 2019
LENTICULAR CLOUDS ~~ NATURAL PHENOMENON
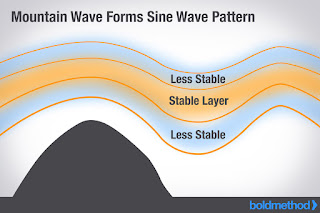
A lenticular cloud is a lens-shaped cloud that normally develops on the downwind side of a mountain or mountain range. This occurs when stable, moist air flows over a mountain, creating a series of oscillating waves. If the temperature at the crest of the wave equals the dew point temperature, condensation occurs in a lens formation. As the air falls down the trough of the wave, where the temperature and dew point temperature are not equal, evaporation occurs. Thus, a wave cloud, or a series of lenticular clouds, is capable of forming. These are often mistaken for UFOs because of the saucer-like shape. They can separate into altocumulus standing lenticular, stratocumulus standing lenticular, and cirrocumulus standing lenticular clouds.
Appearance
How does Lenticular Cloud Form?
As air travels along the surface of the Earth, obstructions are often encountered. These include both natural features of the Earth, such as mountains or hills, and man-made structures, such as buildings and other structures. These disrupt the flow of air into "eddies", or areas of turbulence influenced by these obstructions.
When moist, stable air flows over a larger eddie, like those caused by mountains, a series of large-scale standing waves form on the leeward side of the mountain. If the temperature at the crest of the wave drops below the local dew point, moisture in the air may condense to form lenticular clouds. Under certain conditions, long strings of lenticular clouds can form near the crest of each successive wave, creating a formation known as a "wave cloud." These wave systems can produce large updrafts, occasionally enough for water vapour to condense and produce precipitation.
Where do Lenticular Cloud Form?
Lenticular clouds form in high altitudes, which is why you may have seen
these types of clouds if you live by mountains or you visited
There a frequent occurrence in the Rocky Mountains and places like Mount
Rainier Washington. This can happen as a result of wind shear created by a front.
Are Lenticular Cloud Dangerous?
For most of us lenticular clouds are not dangerous. However pilots do tend
to avoid flying near these flying saucers due to the turbulence to the airplane
systems
Pilots can easily locate the rising air mass based on the orientation of
the clouds making the rough air very easy to spot and thus avoid it.
Pilots of powered aircraft tend to avoid flying near lenticular clouds because of the turbulence of the rotor systems that accompany them, but glider pilots actively seek them out. The precise location of the rising air mass is fairly easy to predict from the orientation of the clouds. "Wave lift" of this kind is often very smooth and strong, and enables gliders to soar to remarkable altitudes and to great distances. As of 2016 the gliding world records for both distance (over 3,000 km; 1,864 mi) and absolute altitude (15,460 m; 50,721 ft) were set using such lift.
Minggu, 24 Maret 2019
Bakti Desa SHS 3 Bandung
The morning sun entered the cold atmosphere in the green of the plantations that lay along the valley, several towering mountains and hills seemed to be the backdrop for the peasants who carried out activities in their fields. With shabby clothes, pants full of mud and soil, hoes, sickles, yellow boots, and typical farmers' hats being their everyday costumes. Without complaining they worked to support themselves and their relatives at home, sometimes their laughter accompanied them to work. Ranging from leaf onion, coffee, carrots, potatoes, cabbage, and tea into a farming community in Cibeureum Village.
PRESENT TO YOU...
BAKTI DESA - Grade XI Students of SHS 3 Bandung City in Cibeureum Village, Kertasari, Bandung Regency.~BACKGROUND~
Village service is an activity that can increase the awareness of students of SMA 3 Bandung towards the residents of Cibeureum Village who have a simple life, and foster sympathy to help one another, at this time.
This village service activity is a form of our concern and social responsibility to be able to provide benefits to the community, especially in the Cibeureum Village Environment. With our passion for serving the community, this village service is a form of assistance.
~PURPOSE OF ACTIVITIES~
1. Build a spirit of solidarity.
2. Increasing caring among others.
3. Carry out responsibilities for our brothers in need.
4. Establishing friendship between humans.
~TIME AND PLACE~
The village service activities are held in Cibeureum Village for 3 days, namely from Thursday, January 3, 2019 until Saturday, January 5, 2019.
~PARTICIPANTS~
This activity was attended by all students - grade XI students of SMA 3 Bandung
~COST~
The total cost for our group in this activity is IDR 1,420,000.00 with the following details:
Transportation Rp. 800,000.00
Consumption of IDR 620,000.00 (IDR 155,000.00 / person)
~CIBEUREUM VILLAGE PROFILE~
EXCELLENCE OF SDA:
1. Cibeureum village is located in a highland area, making it suitable for planting various kinds of plantation and agricultural crops.
2. The main commodities of Cibeureum village are carrots, potatoes, cabbage, coffee and tea.
3. Because of the long distance. This area has cool air.
4. This village has clear and easily available springs.
LIMITATIONS OF SDA:
1. Still lack of processing / utilization of natural resources in Cibeureum village.
~BENEFITS AND LIMITATIONS OF HUMAN RESOURCES~
HR BENEFITS:
1. Actively socializing
2. Friendly and help each other
3. Hardworking and grateful for the situation.
4. Diligently worship
HR LIMITATIONS:
1. The lack of further education facilities.
2. Lack of awareness of waste management.
3. People still believe in mystical things.
4. The community is not yet aware of the importance of driving safety.
~~~JOURNAL OF ACTIVITIES~~~
At 6 o'clock in the morning, grade XI students met in SMA 3 Bandung to go to the destination village. After waiting for 1 hour, we left for Cibeureum village at 7 a.m. We left using public transportation to go there. Each angkot contains fourteen people. We enjoyed our trip by seeing the rice fields and seeing the scenery. We arrived at Cibeureum Village at 9am.
Arriving at the village, we invited at the Cibeureum Village Hall to attend a welcome ceremony from the village head. After that, we had lunch together there. After that, we walked to our homes. By carrying our heavy luggage, we took him walking by comparing meters. We are quite tormented. Finally, after a few minutes we walked, we arrived at our post. After arriving at the post, we went to meet our foster care, Ibu Nyai, and recovered at home.
The feeling of fatigue disappears along with the fresh wind that comes. A glass of water and a lotek plate and also karedok are enough to relieve our fatigue, hunger and fatigue. After eating, we get God's call, which is the call to prayer. We also store our luggage, make prayer tools, then pray at the nearest mosque. After the prayer, we returned to the home of our beloved foster mother.
We talked warmly with Ibu Nyai, then we decided to walk around the village. We went down the village road. In the middle of the trip, we met with other friends and decided to go together to visit the village. We found a soccer field near the Cibeureum Village Hall. At first we wanted to play football there, but Mr. Oman drove us away so we went home with a little regret. We also decided to start fields and gardens and take pictures there. We returned to Azan Asar's compilation house reverberating. We also conducted prayers at the nearest mosque. After the Asar prayer, we proceeded in the opposite direction. We went to the market, then walked through the forest, around the tea garden, down the fields, and looked for a way home. On the way home, we bought and ate ice cream together. We arrived at the house when the sun almost sank in the western horizon.
After taking a break, we carry out Maghrib prayer, then proceed with istighosah. After istighosah, we carry out the Isha prayer, then return to the house. Arriving at home, we ate together in the family room. The food is so delicious and delicious that we add a few servings. After eating, we washed the dishes, then talked with Mrs. Nyai and Mr. Iding. The conversation is very exciting. Mr. Iding told his life story that was very interesting. Not felt, time passed quickly. The clock shows 9pm. We cleaned ourselves, then slept in our room.
Second day, January 4, 2019
At 3:00 in the morning, the dawn was still reluctant to show he was still singing the moon. Our cellphone alarm was awakened by us who was still carried away by a dream. But we were again carried away by the desire to close our eyes in silence. We fell asleep until 5am, then carried out the dawn prayer. After the prayer, we fell asleep again until 7 o'clock, Astagfirullahal adzim. Then we take a shower there. Cold air, cooler water, Brrrrr. Take a bath, we eat and eat with the family in the dining room. As usual, we ate with a heavy voracious appetite. After eating, we wash the dirty dishes ourselves.
Soon, friends from Pak RW's house came to take a walk with beta having classes. Mrs. RW Share my friends and take a walk with us. They carry quite a lot of supplies because it will be cooked very busy.
Our destination is Situ Cisanti. At first, we planned to go walking, but soon the RW RW invited us to go up to the kolungbak buntung. Approximately 25 people climbed into the village. Dangerous indeed, but this is where the class together and parents feel. A 2 km long trip was not felt because of climbing the kolbak. It only takes 10 minutes, we have arrived at the entrance to Situ Cisanti.
Situ Cisanti is a cool and comfortable place to rest and enjoy the sights, especially with family and loved ones. In this place, the air waves from various springs boil down there, known as KILOMETER 0 CITARUM. There we went around and enjoyed the scenery and then ate together.
According to our parents' story, the place was once known as haunted. There are people who have never seen a long snake wrap around a tall tree, a fish the size of 2 doors, even a sighting. There are also animals that are not considered beasts.
Around 10:30, we returned from Situ Cisanti. This time, we walk. The tiring journey felt by almost all the villagers. Crashing through various narrow corners, winding roads, climbing hills, until the caterpillars fell on the shoes, we finally arrived at the house starting at 11:20. We prepare to go to Friday prayers.
We like the Friday prayer usually. In rural areas mosques are scattered in almost every RT. However, only a few Jami mosques are used for Friday prayers. This made the mosque that we occupied full. Some even arrived at the mosque courtyard.
Our lunch in the house is not too big but there is a lot of excitement, namely the home of my beloved Ibu Nyai. As usual, we ate heartily and washed ourselves.
After eating, we went to the village hall to start extension activities, ranging from seeding techniques, tissue culture, plantation and horticulture tools, to workshops to repair damaged tools.
After this activity, we returned to our homes during the Asar Prayer. Pray we at the mosque where the prayer was held on Friday. It seems that the activity on the second day is quite tiring. We rested at the residence that we call the base camp. This is where we share, tell stories and gapleh poker.
Third day, January 5, 2019
The cellphone alarm sounds again. His voice was very noise, so we woke up and went straight to the mosque to carry out the Prayer of the Prophet. After the Prayer prayer, while waiting for breakfast, we went to explore the beautiful plantations. There we saw many farmers who had dripped sweat in the morning to take care of their plants. On the plantation, we also found fish ponds which are used as fishing grounds. When the time showed 7 o'clock, we hurried home to have breakfast and bath. At breakfast, our adoptive mother is very friendly, she serves a lot of food. Very much, one of our friends had to leave first to carry out the student council assignment. After our friend left we all got ready to go to the tea garden with Mr RW.
The journey to the tea garden is very beautiful and spoils the eyes. Before arriving at the tea garden, we stop by at the nursery to see various kinds of garden plant seeds. After that, we continued the journey to the tea garden. Beautiful scenery and cool air welcomes us in the tea garden. The tea plant that was spread out in front of us was very beautiful, so we were tempted to surround the plantation.
On the other hand, our friends who were carrying out their student council assignments were treated to donated books from students of SMAN 3 Bandung. With the books, he will distribute assistance to schools in Cibeureum Village. School after school was visited by him. When the books came, the faces of the school children were visible. They look very happy with books they have never read. The school principals are also happy to accept the donation book. In addition, our friend still has another duty, which is to help the local residents plant trees. There are several trees planted and this is a form of the reforestation movement.
We, the tea garden explorers took many beautiful photos in "LONSUM", that was the call for this tea garden. At 10:30, we all returned to our homes. When we arrive home, we are required to clean up our belongings because soon we will be home to face the reality of school life again.
~~DOCUMENATION~~
Senin, 11 Maret 2019
THIS IS ME
BACKGROUND
HELLO! My name is
Muhamad Fikri Nurohman, people usually call me Fikri.
I was born on the 24th
September 2001, in a city called Sumedang, West Java. So, I'm now 17! I'm the first child in my
family, so I have 1 younger brothers. My younger brother is now 9 years old and
he loves football. My family consists of me, my younger brother, Nabil, my mom,
and my dad.
Although I
was born in Sumedang, I don’t remember that I was lived in Sumedang when I was
child. Since I was born untill my age is 2 years, I lived in a place in
Sumedang called Cariyu. After that, I moved to Bandung, so I don’t remember
anything about Cariyu.
At school, I participate in Trilogi, DKM, Indonesian Debate Forum, and Badminton Club. Trilogi is a group for those who are interested in competing at science stuffs. I'm the representative for the Chemistry group of Trilogi. DKM is an organization to guide me to the right way inlife. Also Indonesian Debate Forum, I learn some softskills in debate, public speaking, and anymore from that. Meanwhile, Badminton club as we all know is a club for those who have interest in Badminton. At that, I play on number MEN’s double with my partner, Gilbert.
WHEN FACE A SPARE TIME :
I like playing games and
watching videos on YouTube.
I really like sports. After a long day at school (at the weekend), I ask my friends to go playing some sports, like football or badminton with me. I think it is the right way to release out stress while also improving my stamina.
Lately, I've been more focused in my studies. I dream to be graduate of my high school and go to the best university I can go to. I'm also preparing for the chemistry olympiads, so these last few months I've been putting extra hours into chemistry so I can have a chance competing in the olympiads.
I really like sports. After a long day at school (at the weekend), I ask my friends to go playing some sports, like football or badminton with me. I think it is the right way to release out stress while also improving my stamina.
Lately, I've been more focused in my studies. I dream to be graduate of my high school and go to the best university I can go to. I'm also preparing for the chemistry olympiads, so these last few months I've been putting extra hours into chemistry so I can have a chance competing in the olympiads.
WHAT IS MY STRENGTH?
Talking about my strengths,
one of them has to be in my ability to learn things quickly. I'm a fast learner
and able to adapt with new things quickly, especially with things that requires
logical things. This greatly affects my grades at school.
When preparing for exams, I don't write notes in books or make beautiful mind maps which consume a lot of time. I just read and understand the concepts which takes a shorter amount of time compared to others who read books and make mind maps, and the results are quite the same, it's usually more effective compared to others.
I'm also pretty good at teaching myself how to do stuff. I can manage my time on how is the way i spend my time. School’s tasks, learn chemistry, doing sports, and pray. I can manage all of it in my life.
I like to help other people too. I don't have a lot of activities so most of the time I'm not busy. On my free time, I usually respond and help the peoples who need my help, usually in school studies or anything, really.
On sports, especially football, I can play almost all in the position. I can be a goalkeeper, a defender, midfielder, and also a striker. When I was young in elementary school, I join Football Association School, and I positioned as a striker. All of my partner believe that I have more skills to be a striker than the others.\
Besides football, I also
like badminton. When palying badminton, I’ve always play with my partner
because I play on men’s double. Although I never join a badminton club before,
I can learn to play quickly, and defeat the others.
MY WEAKNESS
Of course I have a lot of weakness that I have to change it in my life. I just want to tell about my weakness that are most often experienced.
First, I'm often forget something easily. I'm a fast learner, I can memorize something easily, but besides that, I also a forgetful person.
Of course I have a lot of weakness that I have to change it in my life. I just want to tell about my weakness that are most often experienced.
First, I'm often forget something easily. I'm a fast learner, I can memorize something easily, but besides that, I also a forgetful person.
The second is I'm lazy for study about school lesson at home. Sometimes I just imitating my friends homework because I don't like to do a disliked subject. I just want to read and learn about chemistry at home. A lot of e-book stored on my laptop, so I can read it anytime in my spare time, besides to play a game or sports.
Sometimes, I don't care about my body's health, don't care with others. I eat carelessly. I like every food, especially a food that contain cheez on it. But besides that, I also often eat unhealthy foods, so I can say that sometimes, I don't care with my body's health, especially on food selection.
THE THINGS THAT COULD BE
IMPROVED IN ME :
I usually have much free time, and I use them for things that are usually not productive. I spend a lot of time sleeping and playing games when I should have used them for better things I supposed. I spend way too much time on gadgets.
Sometimes I just want to work by myself, not in a group discussion or something, because sometimes It's slow down my work. Besides that, sometimes I also like a group discussion where I can exchange ideas and change my bad perspective. So, you could say – Sometimes I'm a bit selfish on doing my work.
I usually have much free time, and I use them for things that are usually not productive. I spend a lot of time sleeping and playing games when I should have used them for better things I supposed. I spend way too much time on gadgets.
Sometimes I just want to work by myself, not in a group discussion or something, because sometimes It's slow down my work. Besides that, sometimes I also like a group discussion where I can exchange ideas and change my bad perspective. So, you could say – Sometimes I'm a bit selfish on doing my work.
MY ACHIEVEMENTS
Academically, I've lot of achievements. It involves competitions inside school and competitions outside of school, too! At school, I always be a top three at the class. I have a capable skills science.
Academically, I've lot of achievements. It involves competitions inside school and competitions outside of school, too! At school, I always be a top three at the class. I have a capable skills science.
Outside the school, I often join some science competition. Lately, I won the 2nd place in Dentristy Intellectual Challenge with my classmate, Zidane and Aira. It is a national level prestigious competition. The material is Math, physics, biology, chemistry, and basic dendistry knowledge.
I alse have other achievements outside of academics too. When I was a child, I am the 2nd place in Bandung Grassroot Championship. Since that, I continued to practice football until I finished my elementary school. After graduated from elementary school, I join the Junior High School. Since that, I just want to play badminton, but my fooball skills is never lost.
EDU PASSION 2019
Hi guys! How's your day? I hope that you have a nice day. Hmm, I think it's been a long time since the last time I posted. So, can you guest what I'm going to post this time?
24th of January 2019. At that date, my school, 3 Senior High School, held an event named Edu Passion. It is a kind of a fair where universities all around Indonesia promote themselves to attract senior high school students to continue studying there, which is held annually. You can see my video blog of the last year's Edu Passion.This year's Edu Passion is named "Agranasta."
The opening ceremony were held in the morning, participated by the 12th grade students. Ridwan Kamil, our governor, attended it! It was held at the central field where we do our usual ceremonies every Monday.
Around 9am, the real event starts! Stands started opening up, and the whole event was open for public! There were a lot of students from other schools who came to our school just to check out our education fair. It's like an important activity for us because we can take a break from studying and have a moment for us to decide our future: that is, studying at university.
There are so many representatives from many universites around Indonesia that came to SMAN 3 Bandung. They explain about their university briefly, so we can know many information about universities.
Some of the information about the university is there in Bandung and some other cities. I think this event is very useful for students because we can get information as many as we want. We can ask about the university, so we can prepare everything to get there.
For me, Edu Passion is a pretty fun and useful activity to participate in. Not only you can relax from your studies, but also gain a lot of useful information that will surely be used to decide your future (that is if you want to continue your studies to an university).
There are so many representatives from many universites around Indonesia that came to SMAN 3 Bandung. They explain about their university briefly, so we can know many information about universities.
Some of the information about the university is there in Bandung and some other cities. I think this event is very useful for students because we can get information as many as we want. We can ask about the university, so we can prepare everything to get there. Also, there were some performance and food bazaars so we could enjoy the event. I also get a lot of information about how I can get into my favorite university and what I should do to get there.
Here's some photos for during the event:
Senin, 04 Maret 2019
Strength, Weakness, GOALS
My strength
I'm a fast learner
I have skills in many sports
I have a skill in engineering knowledge, like chemistry, i have learned some plane materials because it is very interesting.
Besides, i also like biochem, learn about anatomy, and etc. some knowledge in medical.
My biggest strength is I have Allah
My weakness
I'm stupid, i'm often forget something easily ,
I'm lazy for study about lesson school at home ,
Sometimes, I don't care about my body's health, don't care with others and many more
My goals in future
I want study in bandung institut technology, and go to Germany on my S2
I want to make a plane for Indonesia, just like BJ Habibie.
When i'm successed on want to make a cafe in bandung for the teen chilin in
I want have a harley davidson, lamborghini.
Langganan:
Postingan (Atom)
RECOMMENDATION LETTER
To : Faculty of Electronic and Informatics Engineering Harvard University Dear Dean on Electronic and Informatics Engineering of Harvard...
-
A Conversation between Fikri and Ngux in the Canteen at SMAN 3 Bandung They Talking at the Break Time They're Talking about Plann...
-
Hello! This Time, I would like to share my unforgettable moment in my live When I was in elementary school grade 2, I ever joined a compet...
-
Type of Information : Formal Purpose : To Invite IFA (Iraq Football Association) to attend a meeting at the Home of FIFA (Federation ...